29 results
Length of antibiotic therapy among adults hospitalized with uncomplicated community-acquired pneumonia, 2013–2020
- Natalie L. McCarthy, James Baggs, Hannah Wolford, Sophia V. Kazakova, Sarah Kabbani, Brandon K. Attell, Melinda M. Neuhauser, Lindsey Walker, Sarah H. Yi, Kelly M. Hatfield, Sujan Reddy, Lauri A. Hicks
-
- Journal:
- Infection Control & Hospital Epidemiology , First View
- Published online by Cambridge University Press:
- 14 February 2024, pp. 1-7
-
- Article
- Export citation
-
Objective:
The 2014 US National Strategy for Combating Antibiotic-Resistant Bacteria (CARB) aimed to reduce inappropriate inpatient antibiotic use by 20% for monitored conditions, such as community-acquired pneumonia (CAP), by 2020. We evaluated annual trends in length of therapy (LOT) in adults hospitalized with uncomplicated CAP from 2013 through 2020.
Methods:We conducted a retrospective cohort study among adults with a primary diagnosis of bacterial or unspecified pneumonia using International Classification of Diseases Ninth and Tenth Revision codes in MarketScan and the Centers for Medicare & Medicaid Services databases. We included patients with length of stay (LOS) of 2–10 days, discharged home with self-care, and not rehospitalized in the 3 days following discharge. We estimated inpatient LOT based on LOS from the PINC AI Healthcare Database. The total LOT was calculated by summing estimated inpatient LOT and actual postdischarge LOT. We examined trends from 2013 to 2020 in patients with total LOT >7 days, which was considered an indicator of likely excessive LOT.
Results:There were 44,976 and 400,928 uncomplicated CAP hospitalizations among patients aged 18–64 years and ≥65 years, respectively. From 2013 to 2020, the proportion of patients with total LOT >7 days decreased by 25% (68% to 51%) among patients aged 18–64 years and by 27% (68%–50%) among patients aged ≥65 years.
Conclusions:Although likely excessive LOT for uncomplicated CAP patients decreased since 2013, the proportion of patients treated with LOT >7 days still exceeded 50% in 2020. Antibiotic stewardship programs should continue to pursue interventions to reduce likely excessive LOT for common infections.
National Healthcare Safety Network Antimicrobial Use Option reporting … finding the path forward
- Melinda M. Neuhauser, Amy K. Webb, Arjun Srinivasan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 23 October 2023, e183
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Through the Centers for Medicare and Medicaid Services Promoting Interoperability Program, more hospitals will be reporting to the National Healthcare Safety Network Antimicrobial Use (AU) Option. We highlight the next steps and opportunities for measurement of AU to optimize prescribing.
Description of antibiotic stewardship expertise and activities among US public health departments, 2022
- Destani Bizune, Angelina Luciano, Melinda Neuhauser, Lauri Hicks, Sarah Kabbani
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
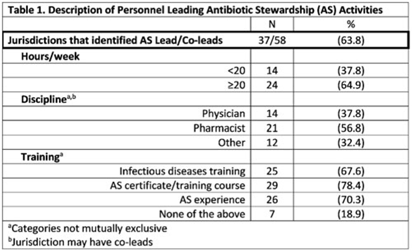
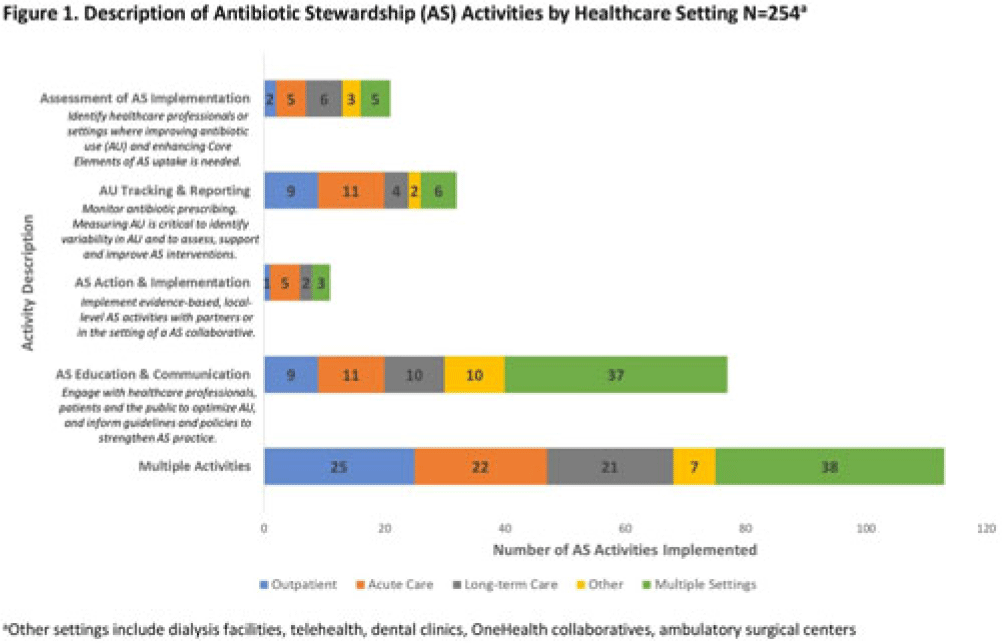
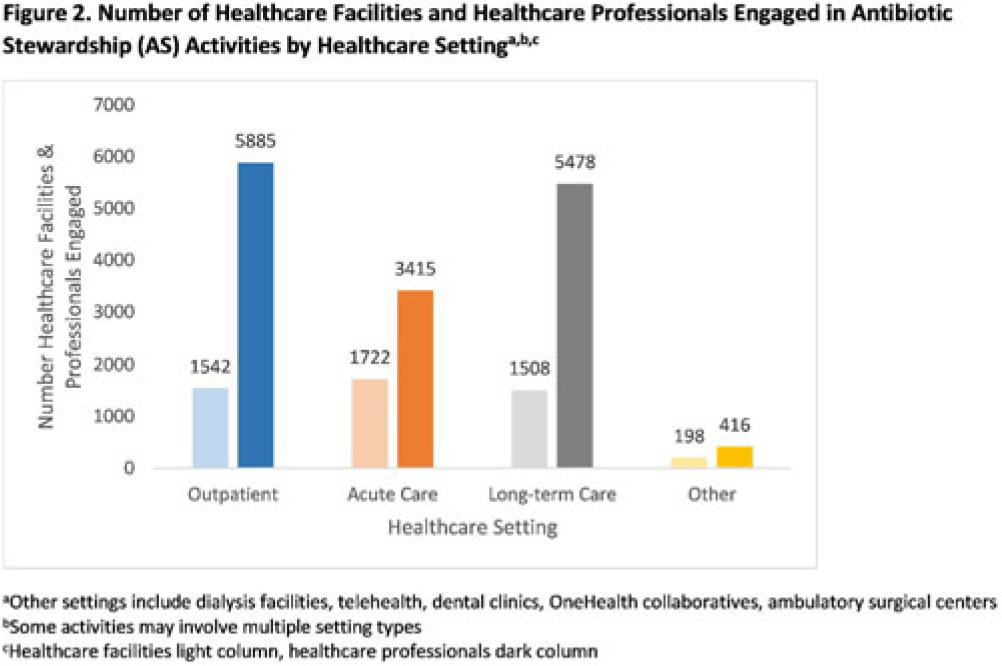
Background: In 2021, the CDC awarded >$100 million to 62 state, local, and territorial health departments (SLTHDs) to expand antibiotic stewardship expertise and implement antibiotic stewardship activities in different healthcare settings. Our objective was to describe SLTHD antibiotic stewardship personnel and activities to characterize the impact of the funding. Methods: SLTHDs submitted performance measures, including quantitative and qualitative responses, describing personnel supporting antibiotic stewardship activities, types of activities, and healthcare facilities and professionals engaged from January through June 2022. A quantitative analysis of performance measures and qualitative thematic analysis of select narrative responses are reported. Results: Most SLTHDs (58 of 62, 94%) submitted performance measures. Among them, 37 (64%) reported identifying an antibiotic stewardship leader or coleader; most were pharmacists (57%) or physicians (38%) with infectious diseases training (68%) (Table 1). Of the remaining STLHDs, 20 reported barriers to identifying a leader or coleader, including hiring process delays and programmatic barriers (Table 2). SLTHDs reported 254 antibiotic stewardship activities; most reported activities involving multiple activity types (44%). Education and communication (eg, providing stewardship expertise) was the most common single activity (30%), followed by antibiotic use tracking and reporting (13%), assessment of antibiotic stewardship implementation (8%), and action and implementation (eg, audit and feedback letters) (4%). The highest number of activities were implemented in multiple healthcare settings (35%), followed by acute care (21%), outpatient (18%), long-term care (17%), and other (9%) (Fig. 1). SLTHDs reported engaging 4,970 healthcare facilities and 15,194 healthcare professionals in antibiotic stewardship activities across healthcare settings, to date, as part of this funding opportunity (Fig. 2). Conclusions: Antibiotic stewardship funding to SLTHDs allowed for increases in capacity and expanded outreach to implement a variety of antibiotic stewardship activities across multiple healthcare settings. Sustaining STLHD antibiotic stewardship activities can help increase engagement and coordination with healthcare facilities, healthcare professionals, and other partners to optimize antibiotic prescribing and patient safety.

Disclosure: None
Empiric antibiotic selection for community-acquired pneumonia in US hospitals, 2013–2020
- Hannah Wolford, Brandon Attell, James Baggs, Sujan Reddy, Sarah Kabbani, Melinda Neuhauser, Lauri Hicks
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s26-s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
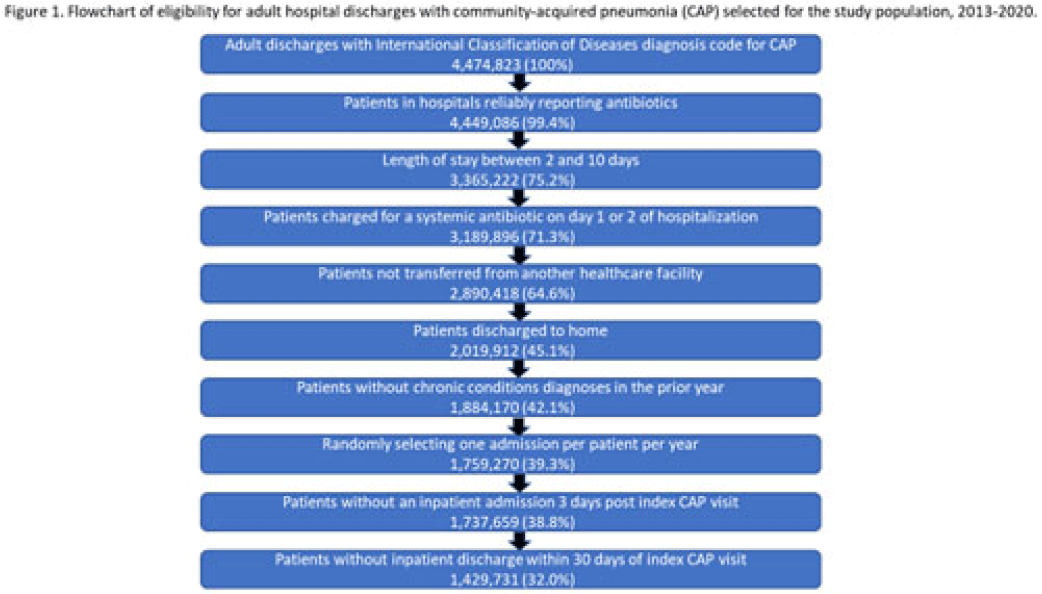
Background: Community-acquired pneumonia (CAP) is a common indication for antibiotic prescribing in hospitalized patients. Professional societies’ clinical guidelines recommend specific antibiotics for empiric treatment of CAP based on clinical factors. Manual assessments of appropriateness are time-consuming and are often conducted on a smaller scale. We evaluated empiric antibiotic selection among a large cohort of adults hospitalized with CAP using electronic health records. Methods: In this study, we used the PINC-AI healthcare database to define a cohort of adults hospitalized with CAP from 2013 to 2020. CAP was identified by International Classification of Diseases (ICD) diagnosis codes. Exclusions were applied to identify uncomplicated CAP (Fig. 1). Treatment was only evaluated if a chest radiograph or computerized tomography (CT) scan was charged during the first 2 days of hospitalization, otherwise it was considered an inadequate CAP evaluation. Administrative billing data were used to identify antibiotics charged within the first 2 days of hospitalization. Empiric guideline-recommended treatment was determined based on 2019 CAP guidelines and more recent studies. Patients who received nonrecommended treatment were evaluated for antibiotic allergies in the current hospitalization or methicillin-resistant Staphylococcus aureus (MRSA) colonization or infection in the year prior or on admission using International Classification of Disease, Tenth Revision (ICD-10) diagnosis codes. Results: We identified 4.47 million adult hospitalizations with CAP from 2013 to 2020; 32% (1.43 million) were included in this analysis (Fig. 1). Among discharges with adequate CAP evaluation (1.37 million), 59.7% received recommended antibiotics in the first 2 days of hospitalization, ranging from 62.6% in 2013 to 57.5% in 2019. Overall, 34.8% of our study population received a nonrecommended antibiotic without documentation of an antibiotic allergy or MRSA colonization (2013: 32.5%; 2018: 36.7%) (Fig. 2). Most patients in our study population received >1 antibiotic (92.3%) in the first 2 days of hospitalization. The most common antibiotics among patients receiving recommended treatment were ceftriaxone (74.2% of patients receiving recommended treatment), azithromycin (67.2%), and levofloxacin (31.8%) (Fig. 3a). The most common nonrecommended antibiotics were vancomycin (57.2% of patients receiving nonrecommended treatment), piperacillin-tazobactam (48.1%), and cefepime (25.7%) (Fig. 3b). From 2013 to 2020, cefepime charges consistently increased among CAP patients treated with nonrecommended antibiotics, whereas levofloxacin charges consistently decreased among CAP patients treated with only recommended antibiotics. Conclusions: Approximately one-third of patients with uncomplicated CAP received nonrecommended empiric antibiotics, and from 2013 to 2020 that proportion increased by 9%. Additional strategies are needed to help identify opportunities to optimize antibiotic selection among patients with CAP.
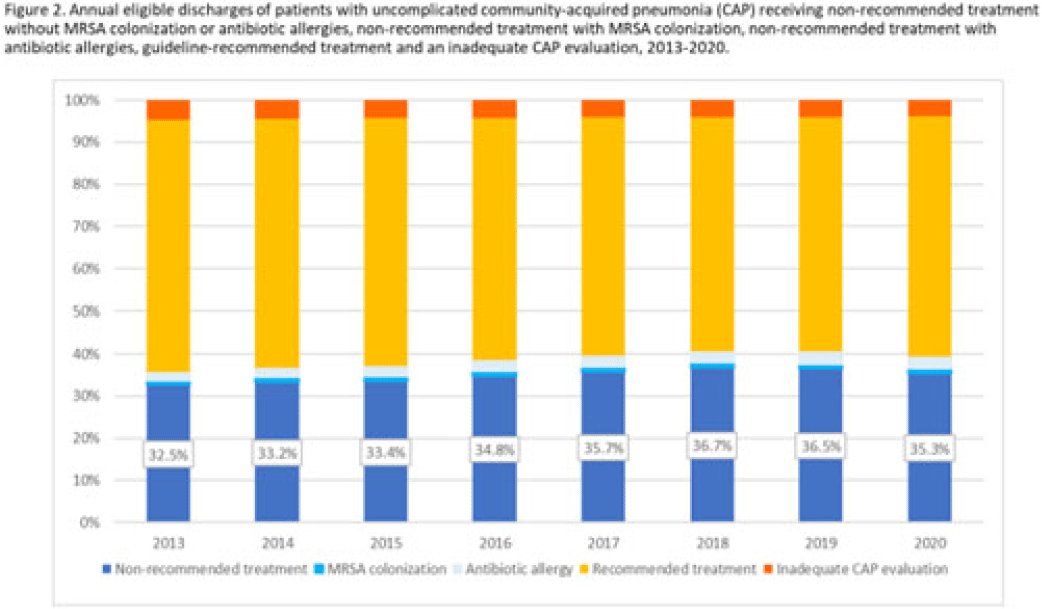
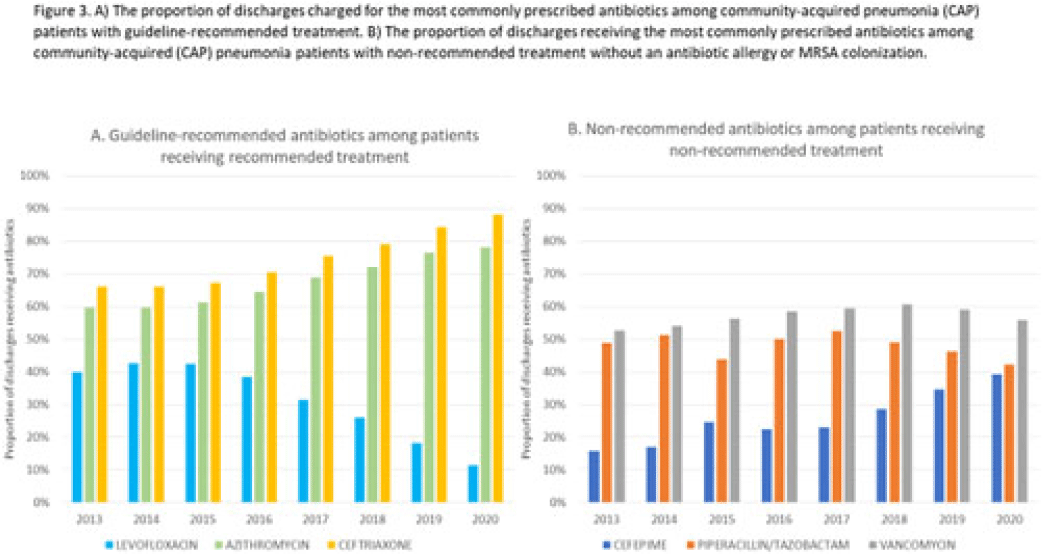
Disclosures: None
Validation of an electronic algorithm to identify appropriate antibiotic use for community-acquired pneumonia in children
- Kathleen Chiotos, Robert Grundmeier, Didien Meyahnwi, Lauren Dutcher, Ebbing Lautenbach, Melinda Neuhauser, Keith Hamilton, Anne Jaskowiak, Leigh Cressman, Julia Szymczak, Brandi Muller, Jeffrey Gerbe
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s106
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Community-acquired pneumonia (CAP) is a common indication for antibiotic use in hospitalized children and is a key target for pediatric antimicrobial stewardship programs (ASPs). Building upon prior work, we developed and refined an electronic algorithm to identify children hospitalized with CAP and to evaluate the appropriateness of initial antibiotic choice and duration. Methods: We performed a cross-sectional study including children 6 months to 17 years hospitalized for CAP between January 1, 2019, and October 31, 2022, at a tertiary-care children’s hospital. CAP was defined electronically as an International Classification of Disease, Tenth Revision (ICD-10) code for pneumonia, a chest radiograph or chest computed tomography scan (CT) performed within 48 hours of admission, and systemic antibiotics administered within the first 48 hours of hospitalization and continued for at least 2 days. We applied the following exclusion criteria: patients transferred from another healthcare setting, those who died within 48 hours of hospitalization, children with complex chronic conditions, and those with intensive care unit stays >48 hours. Criteria for appropriate antibiotic choice and duration were defined based on established guidelines. Two physicians performed independent medical record reviews of 80 randomly selected patients (10% sample) to evaluate the performance of the electronic algorithm in (1) identifying patients treated for clinician-diagnosed CAP and (2) classifying antibiotic choice and duration as appropriate. A third physician resolved discrepancies. The electronic algorithm was compared to this medical record review, which served as the reference standard. Results: Of 80 children identified by the electronic algorithm, 79 (99%) were diagnosed with CAP based on medical record review. Antibiotic use was classified as the appropriate choice in 75 (94%) of 80 cases, and appropriate duration in 16 (20%) of 80 cases. The sensitivity of the electronic algorithm for identifying appropriate initial antibiotic choice was 94%; specificity could not be calculated because no events of inappropriate antibiotic choice were identified based on chart review. The sensitivity and specificity for determining appropriate duration were 88% and 97%, respectively (Table 1).
Conclusions: The electronic algorithm accurately identified children hospitalized with CAP and demonstrated acceptable performance for identifying appropriate antibiotic choice and duration. Use of this electronic algorithm may improve the efficiency of stewardship activities and could facilitate alignment with updated accreditation standards. Future studies validating this algorithm at other centers are needed.
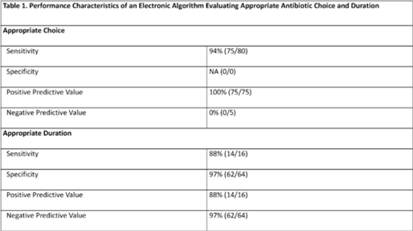
Disclosures: None
Length of antibiotic therapy among adults aged ≥65 years hospitalized with uncomplicated community-acquired pneumonia, 2013-2020
- Natalie McCarthy, Hannah Wolford, Sophia Kazakova, James Baggs, Brandon Attell, Sarah Kabbani, Melinda Neuhauser, Sarah Yi, Kelly Hatfield, Sujan Reddy, Lauri Hicks
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The 2014 US National Strategy for Combating Antibiotic-Resistant Bacteria aimed to reduce inappropriate inpatient antibiotic use by 20% for monitored conditions, such as community-acquired pneumonia (CAP), by 2020. Clinical guidelines recommend treating uncomplicated CAP with a minimum of 5 days of antibiotic therapy. Total length of therapy (LOT) >7 days or >3 days after clinical improvement is rarely necessary. In a previous study estimating LOT in uncomplicated CAP patients, 71% of patients ≥65 years exceeded recommended duration of antibiotics in 2012–2013 (Yi et al, 2018). We evaluated annual trends in LOT in adults ≥65 years hospitalized with uncomplicated CAP from 2013 to 2020. Methods: We conducted a retrospective cohort study among patients in the CMS database with a primary diagnosis of bacterial or unspecified pneumonia using International Classification of Diseases 9th and 10th Revision codes, length of stay (LOS) of 2–10 days, discharged home with self-care, and not rehospitalized in the 3 days following discharge. Discharge home was used as a surrogate for clinical improvement. Because inpatient LOT is not available in CMS data, we used linear regression to model inpatient LOT as a function of LOS using data on CAP patients ≥65 years from the PINC AI healthcare database. Postdischarge LOT was based on prescriptions filled following discharge. Total LOT was calculated by summing estimated inpatient LOT and actual postdischarge LOT (Fig. 1). Total LOT >7 days and postdischarge LOT >3 days were considered indicators of likely excessive LOT. We reported trends in the proportion of patients with likely excessive LOT during the study period. Results: From 2013 through 2020, there were 400,928 uncomplicated CAP hospitalizations among patients aged ≥65 years. Patients were more likely to be female (55%), and they had a median age of 76 years and a median LOS of 3 days. The median total LOT decreased from 9.5 days in 2013 to 7.7 days in 2020. The proportion of patients with total LOT >7 days decreased from 68% in 2013 to 50% in 2020 (% change, −27%); the proportion with postdischarge LOT >3 days decreased from 73% in 2013 to 62% in 2020 (% change, −16%) (Fig. 2). Conclusions: Likely excessive total LOT for adults ≥65 years hospitalized with uncomplicated CAP decreased by 27% in 2020, a considerable improvement from 2013. However, the high proportion of patients with likely excessive postdischarge LOT in 2020 (62%) demonstrates the need for antibiotic stewardship to optimize prescribing at hospital discharge.
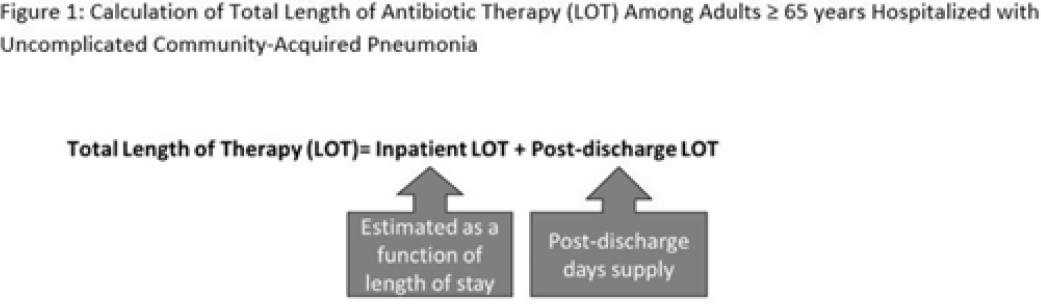
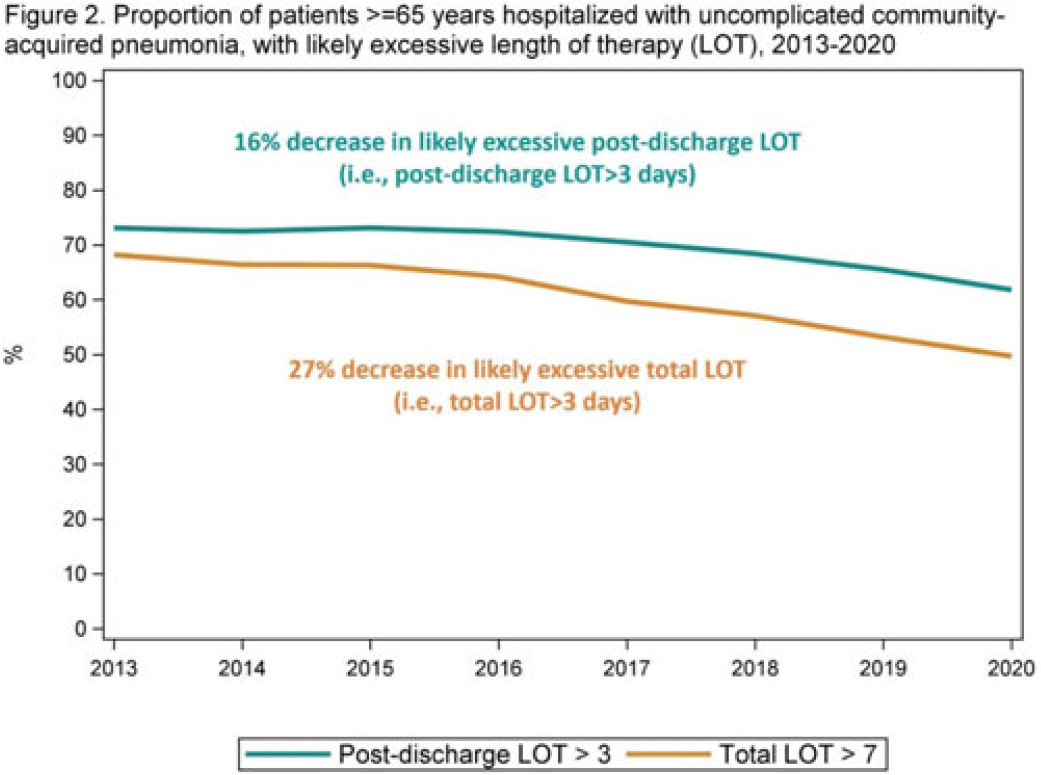
Disclosures: None
Use of leading practices in US hospital antimicrobial stewardship programs
- Edward A. Stenehjem, Barbara I. Braun, Salome O. Chitavi, David Y. Hyun, Stephen P. Schmaltz, Mohamad G. Fakih, Melinda M. Neuhauser, Lisa E. Davidson, Marc J. Meyer, Pranita D. Tamma, Elizabeth S. Dodds-Ashley, David W. Baker
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 6 / June 2023
- Published online by Cambridge University Press:
- 13 October 2022, pp. 861-868
- Print publication:
- June 2023
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Objective:
To determine the proportion of hospitals that implemented 6 leading practices in their antimicrobial stewardship programs (ASPs). Design: Cross-sectional observational survey.
Setting:Acute-care hospitals.
Participants:ASP leaders.
Methods:Advance letters and electronic questionnaires were initiated February 2020. Primary outcomes were percentage of hospitals that (1) implemented facility-specific treatment guidelines (FSTG); (2) performed interactive prospective audit and feedback (PAF) either face-to-face or by telephone; (3) optimized diagnostic testing; (4) measured antibiotic utilization; (5) measured C. difficile infection (CDI); and (6) measured adherence to FSTGs.
Results:Of 948 hospitals invited, 288 (30.4%) completed the questionnaire. Among them, 82 (28.5%) had <99 beds, 162 (56.3%) had 100–399 beds, and 44 (15.2%) had ≥400+ beds. Also, 230 (79.9%) were healthcare system members. Moreover, 161 hospitals (54.8%) reported implementing FSTGs; 214 (72.4%) performed interactive PAF; 105 (34.9%) implemented procedures to optimize diagnostic testing; 235 (79.8%) measured antibiotic utilization; 258 (88.2%) measured CDI; and 110 (37.1%) measured FSTG adherence. Small hospitals performed less interactive PAF (61.0%; P = .0018). Small and nonsystem hospitals were less likely to optimize diagnostic testing: 25.2% (P = .030) and 21.0% (P = .0077), respectively. Small hospitals were less likely to measure antibiotic utilization (67.8%; P = .0010) and CDI (80.3%; P = .0038). Nonsystem hospitals were less likely to implement FSTGs (34.3%; P < .001).
Conclusions:Significant variation exists in the adoption of ASP leading practices. A minority of hospitals have taken action to optimize diagnostic testing and measure adherence to FSTGs. Additional efforts are needed to expand adoption of leading practices across all acute-care hospitals with the greatest need in smaller hospitals.
Tier-based antimicrobial stewardship metrics for genitourinary-related antibiotic use in Veterans’ Affairs outpatient settings
- Matthew Samore, Matthew Goetz, McKenna Nevers, Jacob Crook, Suzette Rovelsky, Ben Brintz, Kelly Echevarria, Melinda Neuhauser, Sharon Tsay, Lauri Hicks, Karl Madaras-Kelly
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s5-s6
-
- Article
-
- You have access Access
- Open access
- Export citation
-
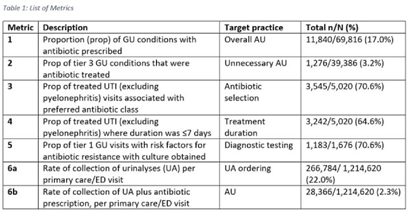
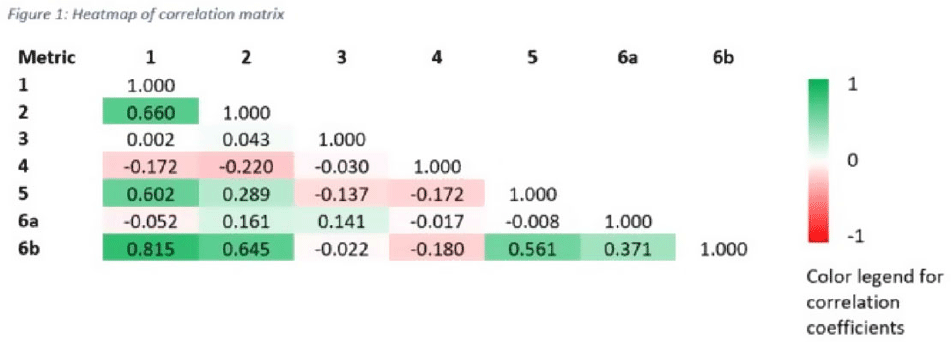
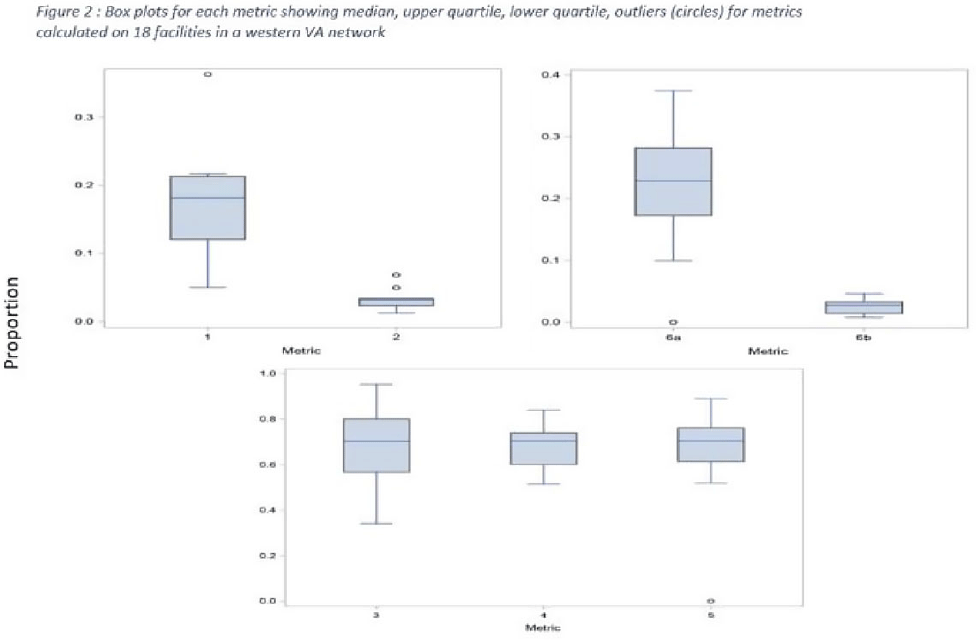
Background: Tracking antibiotic use is a core element of antimicrobial stewardship. We developed a set of metrics based on electronic health record data to support an outpatient stewardship initiative to improve management of urinary tract infections (UTIs) in Veterans’ Affairs (VA) emergency departments (EDs) and primary care clinics. Because UTI diagnostic codes only capture a portion of genitourinary (GU)-related antibiotic use, a tier-based approach was used to evaluate practices. Methods: Metrics were developed to target practices related to antibiotic prescribing and diagnostic testing (Table 1). GU conditions were divided into 3 categories: tier 1, conditions for which antibiotics are usually or always indicated; tier 2, conditions for which antibiotics are sometimes indicated; and tier 3, conditions for which antibiotics are rarely or never indicated (eg, benign prostatic hypertrophy with symptoms). Patients with visits related to urological procedures, nontarget providers, and concomitant non-GU infections were excluded. Descriptive analyses included calculation of the correlation matrix for the 7 metrics and the construction of box plots to display interfacility variability. Results: Metrics were calculated quarterly for 18 VA medical centers, including affiliated clinics, in a western VA network, from July 2018 to June 2020 (Table 1). Tier 3 GU conditions accounted for 1,276 of 11,840 (11%) of GU-related antibiotic use. Metrics 1 and 6b were strongly correlated with each other and were also positively correlated with metrics 2 and 5 (coefficients > 0.5) (Fig. 1). Substantial interfacility variation was observed (Fig. 2). Conclusions: Stewardship metrics for suspected or documented UTIs can identify opportunities for practice improvement. Broadly capturing GU conditions in addition to UTIs may enhance utility for performance feedback. Antibiotic prescribing for tier 3 GU conditions is analogous to unnecessary antibiotic use for acute, uncomplicated bronchitis and upper respiratory tract infections.
Funding: None
Disclosures: None
Patterns of Oral Antibiotic Use and Excess Duration at Hospital Discharge
- Corey Medler, Nicholas Mercuro, Helina Misikir, Nancy MacDonald, Melinda Neuhauser, Lauri Hicks, Arjun Srinivasan, George Divine, Marcus Zervos
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s522-s523
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antimicrobial stewardship (AMS) interventions have predominantly involved inpatient antimicrobial therapy. However, for many hospitalized patients, most antibiotic use occurs after discharge, and unnecessarily prolonged courses of therapy are common. Patient transition from hospitalization to discharge represents an important opportunity for AMS intervention. We describe patterns of antibiotic use selection and duration of therapy (DOT) for common infections including discharge antibiotics. Methods: This retrospective cross-sectional analysis was derived from an IRB-approved, multihospital, quasi-experiment at a 5-hospital health system in southeastern Michigan. The study population included patients discharged from an inpatient general and specialty practice ward on oral antibiotics from November 2018 through April 2019. Patients were included with the following diagnoses: skin and soft-tissue infections (SSTIs), community-acquired pneumonia (CAP), hospital-acquired pneumonia (HAP), respiratory viral infections, acute exacerbation of chronic obstructive pulmonary disease (AECOPD), intra-abdominal infections (IAIs), and urinary tract infections (UTIs). Other diagnoses were excluded. Data were extracted from medical records including antibiotic indication, selection, and duration, as well as patient characteristics. Results: In total, 1,574 patients were screened and 800 patients were eligible for inclusion. The most common antibiotic indications were respiratory tract infections, with 487 (60.9%) patients. These included 165 AECOPD cases (20.6%) and 200 CAP cases (25%) with no multidrug resistant organism (MDRO) risk factors; 57 patients (7.1%) with MDRO risk factors; HAP in 7 patients (0.9%); and influenza in 58 patients (7.2%). Also, 205 (25.6%) patients were diagnosed with UTIs: 71 with cystitis (8.9%), 86 (10.8%) with complicated UTI (cUTI), and 48 (6%) with pyelonephritis. Furthermore, 125 patients (15.6%) were diagnosed with SSTI: 59 (7.4%) purulent and 66 (8.3%) nonpurulent. 31 (3.9%) patients had an IAI. The most commonly used antibiotics were cephalosporins in 536 patients (67%), azithromycin in 252 patients (31.5%), and fluroquinolones and tetracyclines in 231 patients (28.9%). Fluroquinolones were the most frequent antibiotic prescribed at discharge in 210 patients (26.3%). Figure 1 displays the average DOT relative to specific indications. The median duration of total antibiotic therapy exceeded institutional guideline recommendation for multiple conditions, including AECOPD (7 days vs recommended 5 days), CAP with COPD (8.3 vs 7 days ), CAP without COPD (7.7 vs 5 days), and pyelonephritis (11 vs 7–10 days). Also, 269 (33.6%) patients received unnecessary therapy; 218 (27.3%) of these were due to excess duration. Conclusions: Among a cross-section of hospitalized patients, the average DOT, including after discharge, exceeded the optimal therapy for many patients. Further understanding of patterns and influences of antibiotic prescribing is necessary to design effective AMS interventions for improvement.
Funding: This work was completed under CDC contract number 200-2018-02928.
Disclosures: None
Increasing Voluntary Public Health Reporting to the NHSN Antimicrobial Use Option
- Heather Dubendris, Amy Webb, Melinda Neuhauser, Arjun Srinivasan, Wendy Wise, Laura Blum, Erin O’Leary, Jonathan Edwards, Daniel Pollock
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s296-s297
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: The CDC NHSN launched the Antimicrobial Use Option in 2011. The Antimicrobial Use Option allows users to implement risk-adjusted antimicrobial use benchmarking within- and between- facilities using the standardized antimicrobial administration ratio (SAAR) and to evaluate use over time. The SAAR can be used for public health surveillance and to guide an organization’s stewardship or quality improvement efforts. Methods: Antimicrobial Use Option enrollment grew through partner engagement, targeted education, and development of data benchmarking. We analyze enrollment over time and discuss key drivers of participation. Results: Initial 2011 Antimicrobial Use Option enrollment efforts awarded grant Funding: to 4 health departments. These health departments partnered with hospitals, which encouraged vendors to build infrastructure for electronic antimicrobial use reporting. CDC supported vendors through outreach and education. In 2012, with CDC support, Veterans’ Affairs (VA) Informatics, Decision-Enhancement, and Analytic Sciences Center and partners began implementation of Antimicrobial Use Option reporting and validation of submitted data. These early efforts led to enrollment of 64 facilities by 2014 (Fig. 1). As awareness of the antimicrobial use option grew, we focused on facility engagement and development of benchmark metrics. A second round of grant Funding: in 2015 supported submission to the Antimicrobial Use Option from additional facilities by Funding: a vendor, a healthcare system, and an antimicrobial stewardship network. In 2015, CMS recognized the Antimicrobial Use Option as a choice for public health registry reporting under Meaningful Use Stage 3, resulting in an increase in participating hospitals. Antimicrobial Use Option enrollment increased in 2015 (n = 120), coinciding with national prioritization of antimicrobial stewardship. In 2016, the SAAR, was released in NHSN. We leveraged the SAAR to encourage participation from additional facilities and began quarterly calls to encourage continued participation from existing users. In 2016, the Department of Defense began submitting data to the Antimicrobial Use Option, resulting in 207 facilities enrolled in 2016, which grew to 616 in 2017. As of November 2019, 12 vendors self-report submission capabilities and 1,470 facilities, of ~6,800 active NHSN participants, are enrolled in the Antimicrobial Use Option. Two states have passed requirements regulating Antimicrobial Use Option reporting with Tennessee’s requirement going into effect in 2021. Conclusions: The Antimicrobial Use Option offers evidence that collaboration with partners, and leveraging of benchmarking metrics available to a national surveillance system can lead to increased voluntary participation in surveillance of high-priority public health data. Moving forward, we will continue expanding analytic capabilities and partner engagement.
Funding: None
Disclosures: None
Development of an Electronic Algorithm to Identify Inappropriate Antibiotic Prescribing for Pediatric Pharyngitis
- Jeffrey Gerber, Robert Grundmeier, Keith Hamilton, Lauri Hicks, Melinda Neuhauser, Nicole Frager, Muida Menon, Ellen Kratz, Anne Jaskowiak, Leigh Cressman, Tony James, Jacqueline Omorogbe, Ebbing Lautenbach
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s188-s189
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antibiotic overuse contributes to antibiotic resistance and unnecessary adverse drug effects. Antibiotic stewardship interventions have primarily focused on acute-care settings. Most antibiotic use, however, occurs in outpatients with acute respiratory tract infections such as pharyngitis. The electronic health record (EHR) might provide an effective and efficient tool for outpatient antibiotic stewardship. We aimed to develop and validate an electronic algorithm to identify inappropriate antibiotic use for pediatric outpatients with pharyngitis. Methods: This study was conducted within the Children’s Hospital of Philadelphia (CHOP) Care Network, including 31 pediatric primary care practices and 3 urgent care centers with a shared EHR serving >250,000 children. We used International Classification of Diseases, Tenth Revision (ICD-10) codes to identify encounters for pharyngitis at any CHOP practice from March 15, 2017, to March 14, 2018, excluding those with concurrent infections (eg, otitis media, sinusitis), immunocompromising conditions, or other comorbidities that might influence the need for antibiotics. We randomly selected 450 features for detailed chart abstraction assessing patient demographics as well as practice and prescriber characteristics. Appropriateness of antibiotic use based on chart review served as the gold standard for evaluating the electronic algorithm. Criteria for appropriate use included streptococcal testing, use of penicillin or amoxicillin (absent β-lactam allergy), and a 10-day duration of therapy. Results: In 450 patients, the median age was 8.4 years (IQR, 5.5–9.0) and 54% were women. On chart review, 149 patients (33%) received an antibiotic, of whom 126 had a positive rapid strep result. Thus, based on chart review, 23 subjects (5%) diagnosed with pharyngitis received antibiotics inappropriately. Amoxicillin or penicillin was prescribed for 100 of the 126 children (79%) with a positive rapid strep test. Of the 126 children with a positive test, 114 (90%) received the correct antibiotic: amoxicillin, penicillin, or an appropriate alternative antibiotic due to b-lactam allergy. Duration of treatment was correct for all 126 children. Using the electronic algorithm, the proportion of inappropriate prescribing was 28 of 450 (6%). The test characteristics of the electronic algorithm (compared to gold standard chart review) for identification of inappropriate antibiotic prescribing were sensitivity (99%, 422 of 427); specificity (100%, 23 of 23); positive predictive value (82%, 23 of 28); and negative predictive value (100%, 422 of 422). Conclusions: For children with pharyngitis, an electronic algorithm for identification of inappropriate antibiotic prescribing is highly accurate. Future work should validate this approach in other settings and develop and evaluate the impact of an audit and feedback intervention based on this tool.
Funding: None
Disclosures: None
Implementation of Diagnostic Stewardship Algorithms by Bedside Nurses to Improve Culturing Practices: Factors Associated With Success
- Valeria Fabre, Alejandra Salinas, Ashley Pleiss, Elizabeth Zink, George Jones, Lauri Hicks, Melinda Neuhauser, Arjun Srinivasan, Sara Cosgrove
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s276-s277
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Bedside nurses have been recognized as potential antibiotic stewards; however, data on effective ways that nurses can contribute to stewardship activities in acute-care hospitals are scarce. Methods: A nurse-driven urine culture intervention to improve urine culture ordering practices was implemented in a medicine and a neurocritical care unit (NCCU) at The Johns Hopkins Hospital. Bedside nurses implemented an algorithm (Fig. 1) developed by the antibiotic stewardship program (ASP) to review the appropriateness of urine culture and to guide discussions with ordering providers regarding unnecessary urine cultures. Nurses received in-person training by an ASP physician champion on how to use the algorithm and education on the definition and indications for evaluation for asymptomatic bacteriuria and urinary tract infections. The ASP physician periodically visited the units to address concerns and questions. In both units, a nurse champion was identified to serve as liaison between the ASP and bedside nurses, and physician support was obtained before the intervention. The pre- and postintervention periods for the medicine unit were September 2017–August 2018 and September 2018–August 2019, respectively. For the NCCU, these periods were September 2018–February 2019 and March 2019–September 2019, respectively. Trends in urine cultures per 100 patient days (PD) were examined with statistical process charts and compared before and after the intervention using a standard incident ratio (IRR) and Poisson regression. Results: In total, 327 urine cultures were collected in the medicine unit and 293 in the NCCU over the study period. Although the intervention led to a significant 34% reduction in the rate of urine cultures on the medicine unit (from 2.3 to 1.5 cultures/100 PD; IRR, 0.66; 95% CI, 0.50–0.87; P < .01), the number of urine cultures remained without a significant change in the NCCU (from 4.5 to 3.7 cultures/100 PD; IRR, 0.89; 95% CI, 0.65–1.22; P = .48) (Fig. 2). Conclusions: Algorithm-based, nurse-driven review of urine culture indications reduced urine cultures on a medicine unit but not in a neurosciences ICU. Success on the medicine unit may have been driven by highly engaged nurse and physician champions and by patients being able to respond questions about symptoms. The following factors might have impacted results on NCCU: presence of conflicting protocols (eg, panculturing patients every 48 hours per a hypothermia protocol), unit tradition (eg, obtaining cultures to assess treatment response), perception of greater risk benefit in NCCU patients, and unit dynamics (open unit with other primary services placing orders for patients). Unit and team dynamics can affect effective implementation of antimicrobial stewardship interventions by nurses.
Funding: None
Disclosures: None
Hospital Microbiologic Culture Results to Predict the Use of Anti–methicillin-Resistant Staphylococcus aureus (MRSA)
- Hsiu Wu, Tyler Kratzer, Liang Zhou, Minn Soe, Jonathan Edwards, Melinda Neuhauser, Andrea Benin, Lauri Hicks, Arjun Srinivasan, Daniel Pollock
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s40
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: To provide a standardized, risk-adjusted method for summarizing antimicrobial use (AU), the Centers for Disease Control and Prevention developed the standardized antimicrobial administration ratio, an observed-to-predicted use ratio in which predicted use is estimated from a statistical model accounting for patient locations and hospital characteristics. The infection burden, which could drive AU, was not available for assessment. To inform AU risk adjustment, we evaluated the relationship between the burden of drug-resistant gram-positive infections and the use of anti-MRSA agents. Methods: We analyzed data from acute-care hospitals that reported ≥10 months of hospital-wide AU and microbiologic data to the National Healthcare Safety Network (NHSN) from January 2018 through June 2019. Hospital infection burden was estimated using the prevalence of deduplicated positive cultures per 1,000 admissions. Eligible cultures included blood and lower respiratory specimens that yielded oxacillin/cefoxitin–resistant Staphylococcus aureus (SA) and ampicillin-nonsusceptible enterococci, and cerebrospinal fluid that yielded SA. The anti-MRSA use rate is the total antimicrobial days of ceftaroline, dalbavancin, daptomycin, linezolid, oritavancin, quinupristin/dalfopristin, tedizolid, telavancin, and intravenous vancomycin per 1,000 days patients were present. AU rates were modeled using negative binomial regression assessing its association with infection burden and hospital characteristics. Results: Among 182 hospitals, the median (interquartile range, IQR) of anti-MRSA use rate was 86.3 (59.9–105.0), and the median (IQR) prevalence of drug-resistant gram-positive infections was 3.4 (2.1–4.8). Higher prevalence of drug-resistant gram-positive infections was associated with higher use of anti-MRSA agents after adjusting for facility type and percentage of beds in intensive care units (Table 1). Number of hospital beds, average length of stay, and medical school affiliation were nonsignificant. Conclusions: Prevalence of drug-resistant gram-positive infections was independently associated with the use of anti-MRSA agents. Infection burden should be used for risk adjustment in predicting the use of anti-MRSA agents. To make this possible, we recommend that hospitals reporting to NHSN’s AU Option also report microbiologic culture results.
Funding: None
Disclosures: None
Implementation of a Nursing Algorithm for Penicillin Allergy Documentation in the Inpatient Setting
- Valeria Fabre, Alejandra B. Salinas, Lauren Rosales, Arjun Srinivasan, Lauri Hicks, Melinda Neuhauser, Sara Cosgrove
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s270-s271
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Patients with a penicillin/aminopenicillin (PCN) allergy label are more likely to receive non–β-lactam antibiotics and to experience worse clinical outcomes. Given that nurses are often first to interact with patients, we pilot tested a nurse-driven quality improvement initiative to improve PCN allergy documentation and increase β-lactam use. Methods: We conducted a before-and-after study on a labor and delivery unit at The Johns Hopkins Hospital (JHH) from May 2018 to September 2019. Patients aged 18 years with a PCN allergy were included. The intervention included (1) the use of an algorithm developed by the antimicrobial stewardship team to assist nurses in obtaining accurate PCN allergy histories (Fig. 1), (2) identification of a nurse champion to facilitate implementation of the algorithm, and (3) in-person education by a stewardship physician regarding the importance and impact of adequate PCN allergy documentation on clinical outcomes. Readmissions were counted as separate encounters. The primary outcome was improved allergy documentation (either fewer blank documentations, nonspecified rash reactions, drug intolerance documentations (eg, isolated nausea), documentation of signs and symptoms of anaphylaxis not specified as such). The secondary outcome was β-lactam use. Categorical variables were compared using the 2 test and continuous variables were compared with the Student t test. Severe allergic reactions were defined as anaphylaxis, severe skin reactions (eg, Stevens-Johnson syndrome), and organ involvement (eg, hepatitis). Results: Overall, 382 patient admissions were included, 305 in the preintervention (May 2018 to May 2019) and 77 in the postintervention period (June 2019 to September 2019). Mean age and length-of-stay were 30 years and 4 days, respectively, for both periods. The proportion of admitted patients with a PCN allergy label was 8% and 7% for pre- and postintervention periods. Documentation findings in the pre- and postintervention periods respectively were as follows: blank documentation 11% and 12% (P = .89), documentation of specified rashes 0.6% and 1.3% (P = .56), documentation of drug intolerance 11% and 8% (P = .39), documentation of reactions that were indicative of anaphylaxis but not documented specifically as anaphylaxis 8% and 13% (P = .20). Among patients with a documented PCN allergy who received antibiotics, 83 of 177 (47%) and 27 of 43 (63%) received β-lactams (P = .01) in the pre- and postintervention periods, with cefazolin being the antibiotic most commonly used in both periods. Conclusions: Nursing education and an algorithm did not result in significant improvements in PCN allergy documentation in the 3 months after implementation. More data collection is planned to assess the impact of the intervention.
Funding: None
Disclosures: None
Development of an Electronic Algorithm to Target Outpatient Antimicrobial Stewardship Efforts for Acute Bronchitis
- Ebbing Lautenbach, Keith Hamilton, Robert Grundmeier, Melinda Neuhauser, Lauri Hicks, Anne Jaskowiak, Leigh Cressman, Tony James, Jacqueline Omorogbe, Nicole Frager, Muida Menon, Ellen Kratz, Jeffrey Gerber
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s32
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antibiotic resistance has increased at alarming rates, driven predominantly by antibiotic overuse. Although most antibiotic use occurs in outpatients, antimicrobial stewardship programs have primarily focused on inpatient settings. A major challenge for outpatient stewardship is the lack of accurate and accessible electronic data to target interventions. We sought to develop and validate an electronic algorithm to identify inappropriate antibiotic use for outpatients with acute bronchitis. Methods: This study was conducted within the University of Pennsylvania Health System (UPHS). We used ICD-10 diagnostic codes to identify encounters for acute bronchitis at any outpatient UPHS practice between March 15, 2017, and March 14, 2018. Exclusion criteria included underlying immunocompromising condition, other comorbidity influencing the need for antibiotics (eg, emphysema), or ICD-10 code at the same visit for a concurrent infection (eg, sinusitis). We randomly selected 300 (150 from academic practices and 150 from nonacademic practices) eligible subjects for detailed chart abstraction that assessed patient demographics and practice and prescriber characteristics. Appropriateness of antibiotic use based on chart review served as the gold standard for assessment of the electronic algorithm. Because antibiotic use is not indicated for this study population, appropriateness was assessed based upon whether an antibiotic was prescribed or not. Results: Of 300 subjects, median age was 61 years (interquartile range, 50–68), 62% were women, 74% were seen in internal medicine (vs family medicine) practices, and 75% were seen by a physician (vs an advanced practice provider). On chart review, 167 (56%) subjects received an antibiotic. Of these subjects, 1 had documented concern for pertussis and 4 had excluding conditions for which there were no ICD-10 codes. One received an antibiotic prescription for a planned dental procedure. Thus, based on chart review, 161 (54%) subjects received antibiotics inappropriately. Using the electronic algorithm based on diagnostic codes, underlying and concurrent conditions, and prescribing data, the number of subjects with inappropriate prescribing was 170 (56%) because 3 subjects had antibiotic prescribing not noted based on chart review. The test characteristics of the electronic algorithm (compared to gold standard chart review) for identification of inappropriate antibiotic prescribing were the following: sensitivity, 100% (161 of 161); specificity, 94% (130 of 139); positive predictive value, 95% (161 of 170); and negative predictive value, 100% (130 of 130). Conclusions: For outpatients with acute bronchitis, an electronic algorithm for identification of inappropriate antibiotic prescribing is highly accurate. This algorithm could be used to efficiently assess prescribing among practices and individual clinicians. The impact of interventions based on this algorithm should be tested in future studies.
Funding: None
Disclosures: None
Implementation Methods for a Collaborative Pharmacist-Led Antimicrobial Stewardship Intervention at Hospital Discharge
- Corey Medler, Nicholas Mercuro, Nancy MacDonald, Allison Weinmann, Melinda Neuhauser, Lauri Hicks, Arjun Srinivasan, George Divine, Marcus Zervos, Susan Davis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s268-s269
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Unnecessary and prolonged antibiotic use is an important driver of antimicrobial resistance, increasing patient harm and resource utilization. Antimicrobials prescribed at hospital discharge represent an important opportunity to intervene and optimize therapy. Objective: We describe the implementation of a pharmacist-led multidisciplinary antimicrobial stewardship (AMS) intervention at transition of care (TOC) to improve antibiotic selection and duration. Methods: This intervention an IRB-approved multihospital, quasi-experimental, 3-phase stepped-wedge project in a 5-hospital health system. The setting included a large, urban, academic medical center in Detroit, Michigan, and 4 community hospitals in southeastern Michigan. AMS is provided by a pharmacist and infectious diseases physician at each site. For the AMS TOC intervention, pharmacists implemented 3 strategies: (1) early identification of patients to be discharged on oral antibiotics; (2) collaborative planning and communication regarding guideline-recommended antibiotic selection and duration; and (3) facilitation of discharge antibiotic prescription with appropriate stop date. Process improvements were modified to fit the academic and community hospital practice models. The process was implemented in general and specialty practice wards at each hospital site. Prior to implementation in October 2018, pharmacists were trained on tools to standardize identification, collaboration, and documentation. Pocket cards were used to augment education and electronic medical record (EMR) templates standardized documentation. Physicians and nurses on participating units were educated on the rationale and process. Following initiation, ongoing feedback was provided regularly to pharmacists to discuss challenges and to identify solutions. Process measures included the total number of patients receiving the intervention monthly, as indicated by pharmacist AMS TOC notes placed. Protocol adherence was evaluated in 25 randomly selected patients in each study phase each month. Adherence was defined as a pharmacist preparing discharge prescriptions and a placing note in the EMR. Results: Over the study period, 1,558 patient encounters received AMS TOC facilitation by a pharmacist. Monthly protocol adherence ranged from 29% to 87% (higher in academic institutions than community) (Fig. 1). Months of low protocol adherence were associated with times of reduced staffing and onboarding a large group of new employees or trainees. Additional barriers included discharges over weekends. The most common area needing clarification was how to count days of therapy to determine the appropriate stop date. A guide of how to count days of therapy was created to assist. Conclusions: Pharmacist-led antimicrobial stewardship at discharge is a feasible intervention in both academic and community settings. Identifying potential barriers and assessing strategies with multidisciplinary healthcare teams allows for optimal implementation and intervention rollout.
Funding: This work was completed under CDC contract number 200-2018-02928.
Disclosures: None
Improving Prescribing Practices at Hospital Discharge With Pharmacist-Led Antimicrobial Stewardship at Transitions of Care
- Nicholas Mercuro, Corey Medler, Nancy MacDonald, Rachel Kenney, Melinda Neuhauser, Lauri Hicks, Arjun Srinivasan, George Divine, Marcus Zervos, Susan Davis
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s289-s290
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antimicrobial stewardship (AMS) is recommended in hospital, postacute, and outpatient settings. Transitions of care (TOC) are important in each of these settings; however, AMS efforts during TOC have been limited. Beginning in October 2018, we sequentially implemented a pharmacist-led multidisciplinary review of oral antimicrobial therapy prescribed at hospital discharge from general and specialty medicine wards across a health system. Pharmacists facilitated data input of discharge prescriptions following early identification and collaborative discussion of patients to be discharged on oral antimicrobials The purpose of this study was to evaluate the impact of AMS during TOC. Methods: This project was an IRB-approved stepped-wedge, quasi-experimental study in a 5-hospital health system that included hospitalized adults with skin, urinary, intra-abdominal, and respiratory tract infections who had been discharged from general and specialty wards with oral antimicrobials. Patients with complicated infections, neutropenia, or who were transferred from an outside hospital were excluded. The primary end point was optimization of antimicrobial therapy at time of hospital discharge, defined by correct selection, dose, and duration according to institutional guidance. Outcomes were compared before and after the intervention. Results: In total, 800 patients were included: 400 in the preintervention period and 400 in the postintervention period. Among this cohort, 252 (63%) received the intervention by a pharmacist per protocol during TOC. Patients had similar comorbid conditions before and after the intervention. Preintervention patients were more likely to be discharged from community hospitals. Before the intervention, 36% of discharge regimens were considered optimized, compared to 81.5% after the intervention (P < .001); this difference was largely driven by a reduction in patients receiving a duration of therapy beyond the clinical indication (44.5 vs 10%; P < .001). We observed similar clinical resolution, 30-day readmission, and adverse drug events (ADEs) between the pre- and postintervention periods. Postdischarge antimicrobial duration of therapy was reduced from 4 days (range, 3–5) to 3 days (range, 2–4) (P < .001) Severe ADEs occurred more frequently in the preintervention group (9 vs 3.3%; P = .001), which was driven by isolation of multidrug-resistant pathogens (7 vs 2.5%; P = .003) and Clostridioides difficile (1.8 vs 0.5%; P = .094). Patients who received optimal therapy at discharge were less likely to develop an ADE (aOR, 0.530; 95% CI, 0.363–0.773). Conclusions: Implementation of an AMS TOC protocol reduced antimicrobial days, optimized therapy selection, and reduced duration. This intervention was associated with improved safety without compromise of clinical effectiveness. To increase patient safety, AMS programs should target antimicrobial optimization during TOC.
Funding: This work was completed under CDC contract number 200-2018-02928.
Disclosures: None
Development of an Electronic Tool to Measure Daily Appropriateness of Inpatient Antibacterial Use
- Vanessa Stevens, Pamela Belperio, Melinda Neuhauser, Lauri Hicks, McKenna Nevers, Matthew Goetz, Nai-Chung Chang, Arjun Srinivasan, Makoto Jones
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s2
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Assessing antimicrobial use (AU) appropriateness isa cornerstone of antimicrobial stewardship, largely accomplished through time-intensive manual chart review of specific agents or diagnoses. Efforts to evaluate appropriateness have focused on assessing the appropriateness of an entire treatment course. An electronic measure was developed to assess the appropriateness of each day of inpatient AU leveraging electronic health record data. Methods: We extracted contextual data, including risk factors for resistant organisms, allergies, constitutional signs and symptoms from diagnostic and procedural codes, and microbiological findings, from the electronic health records of patients in Veterans’ Health Administration inpatient wards reporting data to the National Healthcare Safety Network (NHSN) AU option from 2017–2018. Only the antibacterial categories shown in Figure 1 were included. Respiratory, urinary tract, skin and soft-tissue, and other infection categories were defined and applied to each hospital day. Algorithm rules were constructed to evaluate AU based on the clinical context (eg, in the ICU, during empiric therapy, drug–pathogen match, recommended drugs, and duration). Rules were drawn from available literature, were discussed with experts, and were then refined empirically. Generally, the rules allowed for use of first-line agents unless risk factors or contraindications were identified. AU was categorized as appropriate, inappropriate, or indeterminate for each day, then aggregated into an overall measure of facility-level AU appropriateness. A validation set of 20 charts were randomly selected for manual review. Results: Facility distribution of appropriateness, inappropriateness, and indeterminate AU by 4 of the adult, 2017 baseline NHSN Standardized Antimicrobial Administration Ratio (SAAR) categories are shown in Figure 1. The median facility-level inappropriateness across all SAAR categories was 37.2% (IQR, 29.4%–52.5%). The median facility-level indeterminate AU across all SAAR categories was 14.4% (IQR, 9.1%–21.2%). Chart review of 20 admissions showed agreement with algorithm appropriateness and inappropriateness in 95.4% of 240 antibacterial days.
Conclusions: We developed a comprehensive, flexible electronic tool to evaluate AU appropriateness for combinations of setting, antibacterial agent, syndrome, or time frame of interest (eg, empiric, definitive, or excess duration). Application of our algorithm in 2 years of VA acute-care data suggest substantial interfacility variability; the highest rates of inappropriateness were for anti-MRSA therapy. Our preliminary chart review demonstrated agreement between electronic and manual review in >95% of antimicrobial days. This approach may be useful to identify potential stewardship targets, in the development of decision support systems, and in conjunction with other metrics to track AU over time.
Funding: None
Disclosures: None
Evaluation of uncomplicated acute respiratory tract infection management in veterans: A national utilization review
- Jefferson G. Bohan, Karl Madaras-Kelly, Benjamin Pontefract, Makoto Jones, Melinda M. Neuhauser, Matthew Bidwell Goetz, Muriel Burk, Francesca Cunningham, for the ARI Management Improvement Group
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 40 / Issue 4 / April 2019
- Published online by Cambridge University Press:
- 11 April 2019, pp. 438-446
- Print publication:
- April 2019
-
- Article
- Export citation
-
Background:
Antibiotics are overprescribed for acute respiratory tract infections (ARIs). Guidelines provide criteria to determine which patients should receive antibiotics. We assessed congruence between documentation of ARI diagnostic and treatment practices with guideline recommendations, treatment appropriateness, and outcomes.
Methods:A multicenter quality improvement evaluation was conducted in 28 Veterans Affairs facilities. We included visits for pharyngitis, rhinosinusitis, bronchitis, and upper respiratory tract infections (URI-NOS) that occurred during the 2015–2016 winter season. A manual record review identified complicated cases, which were excluded. Data were extracted for visits meeting criteria, followed by analysis of practice patterns, guideline congruence, and outcomes.
Results:Of 5,740 visits, 4,305 met our inclusion criteria: pharyngitis (n = 558), rhinosinusitis (n = 715), bronchitis (n = 1,155), URI-NOS (n = 1,475), or mixed diagnoses (>1 ARI diagnosis) (n = 402). Antibiotics were prescribed in 68% of visits: pharyngitis (69%), rhinosinusitis (89%), bronchitis (86%), URI-NOS (37%), and mixed diagnosis (86%). Streptococcal diagnostic testing was performed in 33% of pharyngitis visits; group A Streptococcus was identified in 3% of visits. Streptococcal tests were ordered less frequently for patients who received antibiotics (28%) than those who did not receive antibiotics 44%; P < .01). Although 68% of visits for rhinosinusitis had documentation of symptoms, only 32% met diagnostic criteria for antibiotics. Overall, 39% of patients with uncomplicated ARIs received appropriate antibiotic management. The proportion of 30-day return visits for ARI care was similar for appropriate (11%) or inappropriate (10%) antibiotic management (P = .22).
Conclusions:Antibiotics were prescribed in most uncomplicated ARI visits, indicating substantial overuse. Practice was frequently discordant with guideline diagnostic and treatment recommendations.
Index
- Edited by Tamar F. Barlam, Melinda M. Neuhauser, Pranita D. Tamma, The Johns Hopkins University School of Medicine, Kavita K. Trivedi
-
- Book:
- Practical Implementation of an Antibiotic Stewardship Program
- Published online:
- 06 April 2018
- Print publication:
- 26 April 2018, pp 322-332
-
- Chapter
- Export citation
















